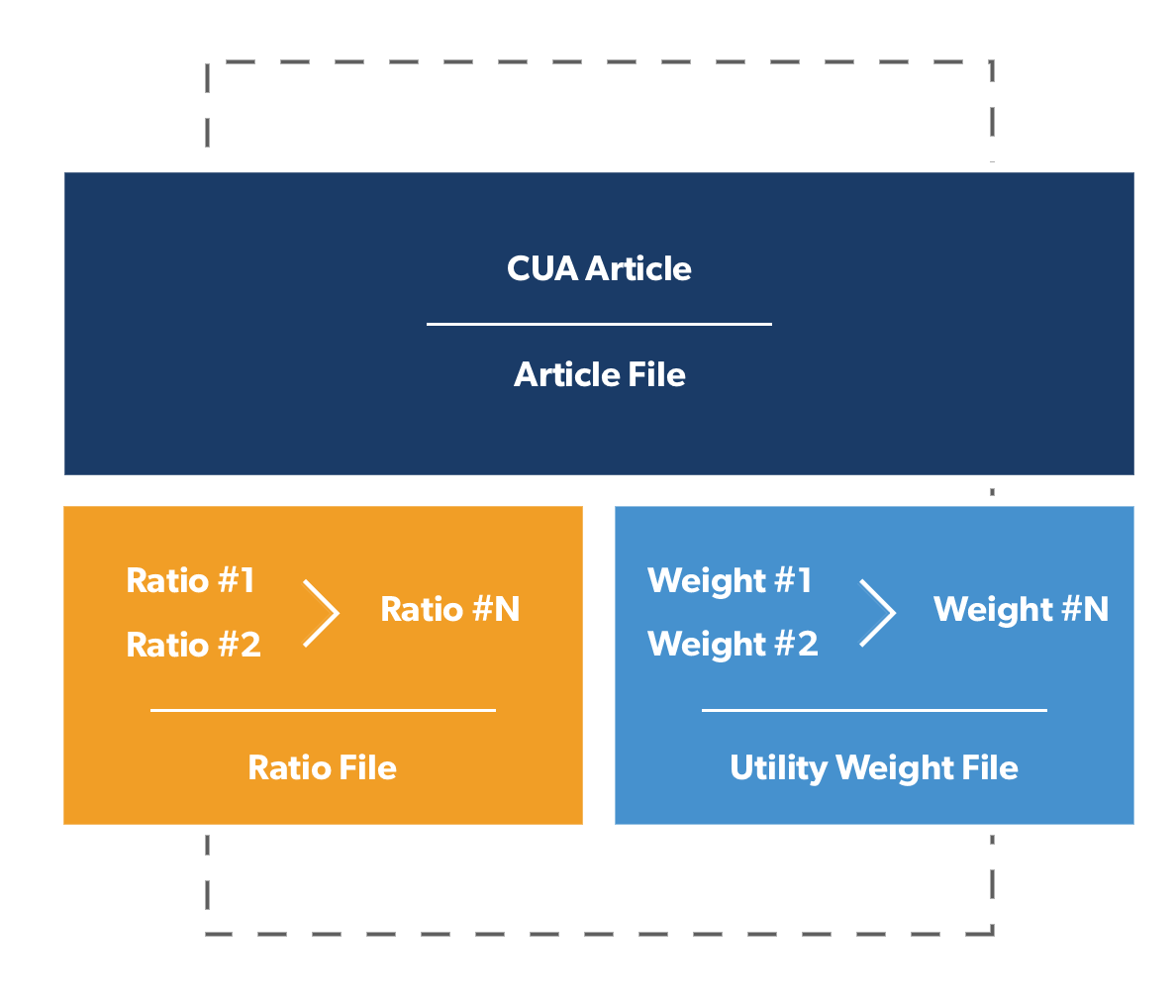
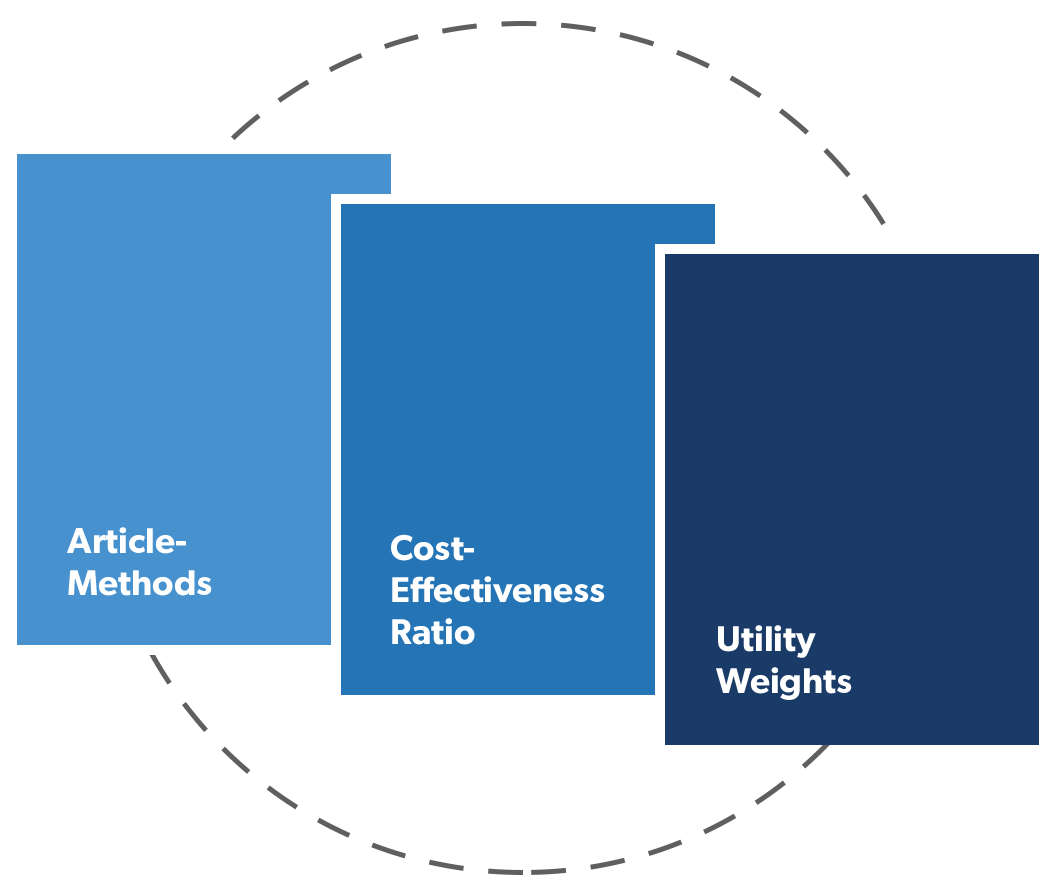
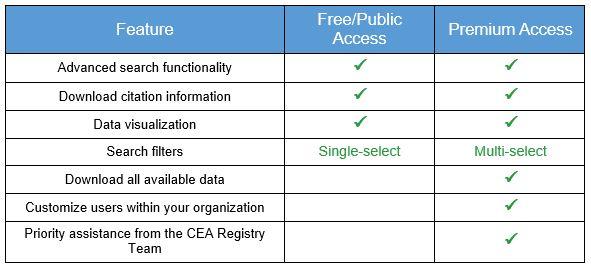
Papers that satisfy our inclusion criteria must:
- Be an original cost-effectiveness analysis
- Measure health benefits as QALYs
If you believe a paper satisfies these criteria and should be considered, let us know.
Excluded articles include the following:
- Systematic reviews
- Opinion/perspective/editorial/commentaries
- Methodological article
- Health Technology Assessments (HTAs)
- Study protocols
- Cost benefit analysis (CBA)
- CEAs that measure health benefits using other metrics besides QALYs or DALYs (e.g., LYs, HALYs, cases)
- Other non-economic analysis